Methods
The proposed project plans a series of short-term laboratory experiments on the effects of seawater acidification (increased CO2 level) on geochemical processes and biota using an unique hyperbaric tank, the so called Karl Erik TiTank. Surface sediments and two macrofaunal species, which dominate in the area of potential CO2 storgae site in the southern Baltic Sea (B3 field) will be sampled and transported wet and cool to the Norwegian partner (SINTEF) for exposure experiments.
The Karl Erik TiTank was developed by the Norwegian University of Science and Technology, Trondheim (NTNU), SINTEF, Trondheim and Statoil to simulate CO2 leakage scenarios under controlled, yet realistic conditions in laboratory. The hyperbaric tank is a flow-through chamber having a total volume of 1.4 m3 which can attain a pressure of up to 30 bars (corresponding to approximately 310 m water depth). Increase in seawater acidity can be simulated by maintaining CO2 concentration at a given level while at constant water flow rate, hydrostatic pressure and temperature. The tank allows thus to simulate conditions at the potential storage site in the Baltic Sea. The tank is constructed from titanium which is not affected by the corrosive properties of seawater at the relevant physical conditions, ensuring no contamination from the tank material during the experiments. The tank is equipped with light source (visible and IR) and a robotic arm with an attached camera. Samples can be collected through a decompression chamber without reducing the pressure in the tank. Addition of gaseous CO2 is regulated by highly accurate mass flow controllers. The maximum seawater flow through rate is 1.0 L min-1 and a maximum recirculation rate is 16.0 L min-1. Water and sediment samples can be continuously collected from the tank during experiments. A six-channel micro sampling system is under development to permit collection of blood samples from animals and/or pore water from sediments during experimental runs in the tank.
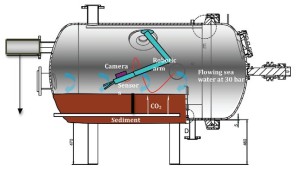
Transect of Karl Erik TiTank (Ardelan et al., 2012)
In the pressurised TiTank, sediments and macrobenthic faunal species which are dominant in the B3 field will be exposed over 30 days to different CO2 levels which will simulate a range water alkalinity gradient as distance from the leak increases. During the course of the experiment the following geochemical processes and biological responses will be investigated to assess environmental risk due to CO2 leakage:
a) Chemical and geochemical processes
1. Acidification rate and bubble formation at different CO2 levels in seawater at pressures relevant to seabed conditions (80 m)
2. Measurements of the concentration and speciation of metals released in sediments/pore water as well as metals released to the seawater under different CO2 conditions at hydrostatic pressures relevant for seabed conditions (9 bar).
3. Assessment of low pH-driven dissolution of apatite and non-apatite P in the Baltic sediments and pore water.
b) Numerical modelling
Application of the modified redox-processes transport reaction O-C-N-P-Si-S-Fe-Mn-Si model adopted for the sediment water interface (the Bottom RedOx Model, BROM) for:
1. analyses of the experimental data,
2. calculation of distributions and fluxes.
3. evaluation consequences of the different scale leakages on the sediment-water interface biogeochemistry
c) Biological impacts
1. Functional and structural responses of benthic communities to elevated CO2 levels, including assessment of qualitative and quantitative changes of macro- and meiobenthic communities and net system respiration.
2. Qualitative and quantitative effects of different CO2 levels on key benthic species, i.e. one calcifying species such as the Baltic clam Macoma balthica (Bivalvia) and one non-calcifying species such as a ragworm Hediste diversicolor (Polychaeta). These two species dominate benthic communities in deeper Baltic Sea and therefore can serve as a good model key-species. If feasible, we propose to conduct studies on both, adult and larval life stages that will address responses at different life stages.
3. Measurements of CO2 buffering capacity that possibly translates into greater tolerance to ocean acidification. Marine invertebrates have evolved three basic mechanisms to counteract the effects of metabolically produced CO2 and hydrogen ion: (i) passive buffering, (ii) active CO2 transport and ion exchange, and (iii) metabolic suppression (Fabry et al. 2008). The same mechanisms are used for neutralizing environmental acidification, hence it can be speculated that species, which are more tolerant to acidosis and (high CO2), show greater capacity to counteract disturbance of acid-base status (Seibel and Walsh 2003). To test these hypothesis, biomarkers such as decalcification rate and carbonic anhydrase activity will be used as a proxy of acid-base regulation capacity.
4. Selection of community, species-level and physiological biomarkers of elevated CO2 for environmental monitoring above the CO2 sub-seabed storage site.
 Polski
Polski